Significance Statement
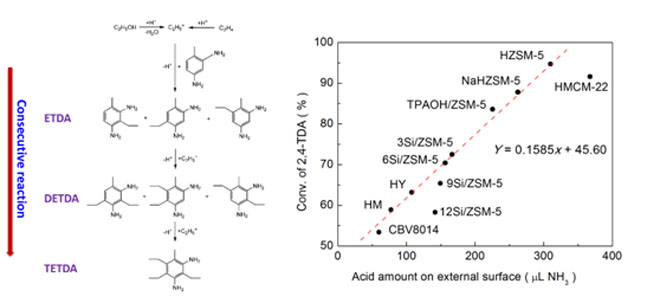
Diethyl toluene diamine (DETDA) is an important curing agent of polyurethane and epoxy resin. The current industrial process for Diethyl toluene diamine manufacture mainly employs the environmentally hazardous Lewis acids such as aluminum alkyls as the catalyst for 2,4-toluene diamine (2,4-TDA) ethylation. A novel route for the environmentally friendly synthesis of Diethyl toluene diamine was developed by the ethylation of 2,4-TDA over acidic zeolites using ethene and ethanol as the alkylation reagents. Different types of zeolites and reaction conditions were examined. The ethylation of 2,4-TDA occurred mainly on the external surface of zeolites, due to the diffusion limitation of pore diameter. The acidity, especially the Brønsted acid sites on the external surface of zeolites, plays a more important role in the ethylation than the pore channel does. The treatment with NaOH generating meso- and macropores and increasing external surface area can also improve the catalytic activity of ZSM-5. When the reaction was carried out in a batch reactor at 330 °C for 5 h using ethanol as the ethylation reagent, the HZSM-5 treated with 0.6 mol/L NaOH showed the best catalytic performance (XTDA, SETH2 and SETH22 reached to 98.4%, 47.4% and 58.8%, respectively).
Journal Reference
Ind. Eng. Chem. Res., 2015, 54 (30), pp 7364–7372.
Yi Zuo1, Xiaowa Nie1, Min Liu1, Ting Zhang1, Chengyi Dai1, Fanshu Ding1, Chunshan Song2, Xinwen Guo1
[expand title=”Show Affiliations”]- State Key Laboratory of Fine Chemicals, PSU-DUT Joint Center for Energy Research, Department of Catalysis Chemistry and Engineering,Dalian University of Technology, Dalian 116024, People’s Republic of China
- EMS Energy Institute, PSU-DUT Joint Center for Energy Research and Department of Energy & Mineral Engineering,Pennsylvania State University, University Park, Pennsylvania 16802,United States [/expand]
Abstract
A novel route for the environmentally friendly synthesis of diethyl toluene diamine (DETDA) was developed by the ethylation of 2,4-toluene diamine (2,4-TDA) over acidic zeolites using ethene and ethanol as the alkylation reagents. Different types of zeolites and reaction conditions were examined. Monoethyl toluene diamine (ETDA) was first generated and then Diethyl toluene diamine was formed by ethylation of ETDA. Triethyl toluene diamine (TETDA) was the excessive ethylation product. The acidic zeolites were characterized by NH3 temperature-programmed desorption, pyridine adsorption Fourier transform infrared spectroscopy, argon/nitrogen physisorption and scanning/ transmission electron microscopy. The acidity, especially the Brønsted acid sites on the external surface of zeolites, plays a more important role in the ethylation than the pore channel does. A small crystal size exposing more acid sites on the external surface is beneficial for the producing of DETDA. The treatment with NaOH generating meso- and macropores and increasing external surface area can also improve the catalytic activity of ZSM-5.
Copyright © 2015 American Chemical Society.
Go To Ind. Eng. Chem. Res
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.


