Significance Statement
Titanium dioxide has received much research attention in many areas of science and technology owing to its attractive applications in dielectrics, photo catalysts, bio applications, water splitting, and energy applications. For this reason, nanostructured titanium dioxide with varying morphologies such as 0-2 dimensional structures has been produced following various procedures. Surface functionalization of titanium oxide has a positive efficacy in various biomedical applications including implants, drug delivery, and bone scaffolds.
However, there has been a challenge of clearly understanding the bonding strength at the interfaces and resistance of the thin film substrate systems. This arise from the high atomic coordination at the face between the substrate and the films. Tantalum has high bioactivity and corrosion resistance as compared to other biocompatible elements. However, the high cost of tantalum and bulk density, limits its clinical application potential.
A microlayer of tantalum embedded on the surface of titanium can enhance corrosion and bioactivity. Adding antibacterial ions of silver or gold can reduce critical microbial infection. For this reason, doping antibacterial agent ions of silver or gold can enhance the antibacterial capability of tantalum-coated titanium alloy. Jalal Azadmanjiri and colleagues presented a simple electrostatic polarization method in an electronic model that helped them investigate the interfacial bonding between tantalum or tantalum silver thin films and titanium alloy substrates. Their work is published in journal, industrial and Engineering chemistry research.
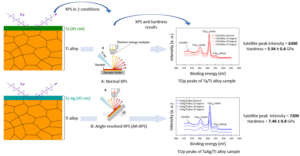
The authors used X-ray photoelectron spectroscopy to assess the interfaces of Tantalum-silver/titanium alloys and tantalum/titanium alloy systems. Their main problem was to identify the electronic structure of the conjugated film on the metal surface. Therefore, they adopted an X-ray photoelectron spectroscopy to assess (i) tantalum coated titanium alloy and (ii) tantalum-silver coated titanium alloy systems with an aim of capturing the satellite peak feature of the titanium 2p photoelectron line. The peaks were greatly influenced by valence electrons as well as electron density distribution. Therefore, the authors used peak intensity as a direct measure of the valence electron density.
The authors obtained results that were consistent with the x-ray photoelectron spectroscopy data for heterostructure specimens. They recorded an interfacial polarization from the X-ray photoelectron spectroscopy spectra which indicated an enhanced bonding between titanium atoms at the interface. When they increased the bonding coordination and likely interstitial and diffusions of silver with atomic radii less than tantalum and titanium, created denser and stiffer coatings. They realized that these coatings limited the conventional movements of interfacial dislocations, therefore, increasing the hardness of the obtained surface system.
This study investigated the interfacial bonding structures of tantalum-silver and tantalum films on titanium surfaces using electronic models, x-ray photoelectron spectroscopy, and nanoindentation. They realized that satellite peak intensity had a direct relationship with bonding strength as well as the hardness of the surface. The bonding strength of the film was further enhanced by the formation of semiconductor phases with large band gaps at the tantalum-silver and titanium alloys.
Reference
Jalal Azadmanjiri1, James Wang1, Christopher C. Berndt1,2, Ajay Kapoor1, De Ming Zhu1, Andrew S. M. Ang1, and Vijay K. Srivastava3. Tantalum- and Silver-Doped Titanium Dioxide Nano sheets Film: Influence on Interfacial Bonding Structure and Hardness of the Surface System. Industrial and Engineering chemistry research, volume 56 (2017), pages 434−439.
[expand title=”Show Affiliations”]- School of Engineering, Faculty of Science, Engineering and Technology, Swinburne University of Technology, Hawthorn, Victoria 3122, Australia
- Department of Materials Science and Engineering, Stony Brook University, Stony Brook, New York 11794, United States
- Department of Mechanical Engineering, Indian Institute of Technology, BHU, Varanasi-221005, India
Go To Industrial and Engineering chemistry research
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.








