Significance
The meteoric rise of lithium-ion batteries has reshaped how we store and deliver energy and they are found in nearly every aspect of modern infrastructure from our smartphones to massive electric vehicle fleets and grid-scale storage. But what tends to get far less attention is what happens once these batteries are spent. That part is becoming harder to ignore. Beyond the obvious e-waste problem, used LIBs carry significant economic potential. They’re dense with valuable metals—lithium, cobalt, nickel, manganese—all of which are increasingly expensive and geopolitically complicated to source. Recycling offers a pathway forward, but most existing methods are far from ideal. The dominant industrial approach today relies on hydrometallurgy—basically dissolving battery materials in strong acids to extract the metals. It’s effective, yes, but costly and chemically messy. It produces considerable liquid waste, and separating lithium from the cocktail of other metals remains frustratingly inefficient. The economics get even murkier when you factor in the cleanup. Dry processing techniques have been floated as an alternative. They’re cleaner on paper—no acids, no water treatment—and they align better with high-temperature industrial setups. But so far, they’ve struggled with finesse. Separating lithium without also losing or contaminating the transition metals has proven extremely tricky. Thermal routes often volatilize everything at once, which defeats the point.
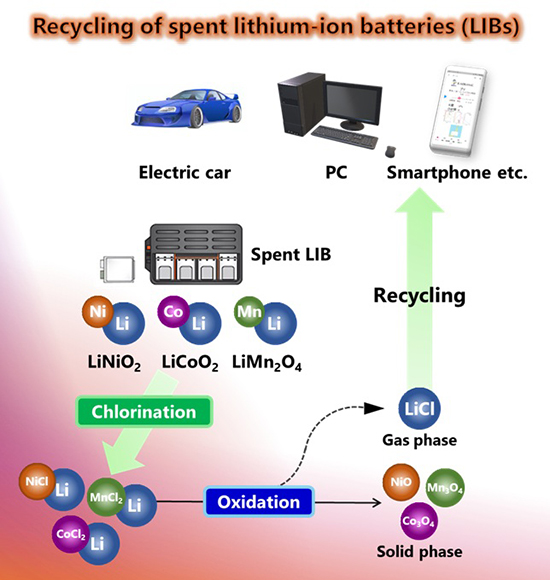
To this account, new research paper published in Reaction Chemistry & Engineering and led by Assistant Professor Yuuki Mochizuki and Associate Professor Naoto Tsubouchi from the Faculty of Engineering at Hokkaido University explored a new smarter dry route. Their approach is refreshingly straightforward, but chemically well-considered. They took three common cathode chemistries—LiNiO₂, LiCoO₂, and LiMn₂O₄—and treated them first with chlorine gas at moderate temperatures, enough to form metal chlorides without triggering early volatilization. Then, in a second thermal step, they oxidized the chlorinated materials in air at high temperatures. Lithium chloride, being both volatile and oxidation-resistant, evaporated away cleanly. The transition metals, in contrast, converted into solid oxides—NiO, Co₃O₄, Mn₂O₃—that stayed behind.
The authors started by testing how three typical cathode compounds—LiNiO₂, LiCoO₂, and LiMn₂O₄—behaved when exposed to chlorine gas under a range of temperatures. At first, it might’ve seemed like a matter of simply heating and watching the metals volatilize one by one. But reality, as always, proved less cooperative. Once the temperature crept past about 700 °C, all four metals—lithium included—began to volatilize more or less in tandem. That made any kind of selective separation basically impossible. Below that point, though, they noticed something different. The metals reacted with chlorine to form chlorides, but didn’t immediately volatilize. XRD scans confirmed this, revealing peaks for LiCl, NiCl₂, CoCl2, and MnCl2. So they had a chemically distinct mixture—but it wasn’t yet separating on its own. To tip the balance, they added a second step: heating these chlorinated samples in air. And here’s where things got more interesting. Lithium chloride, which doesn’t oxidize easily and has a relatively low boiling point, started volatilizing rapidly once the temperature hit 800 °C. By 1300 °C, more than 95% of the lithium had evaporated. The other metals—Ni, Co, and Mn—behaved quite differently. Their chlorides oxidized and settled into solid oxides like NiO, Co₃O₄, and Mn2O3, which remained in the residue. That contrast—between what vanished and what stayed—was the crux of the strategy. The researchers afterward ran thermogravimetric and XRD analyses and confirmed the transformations. No detectable chloride phases remained for the transition metals post-oxidation. It wasn’t just lithium leaving—it was doing so on thermodynamic terms that left the others untouched. To stress-test their method, they compared it to the standard wet approach (chlorination followed by water leaching). The differences were pretty stark. Lithium recovery jumped to nearly 100% in their system, while the other metals barely budged. What’s more, kinetic modeling showed lithium volatilized at a significantly lower activation energy, reinforcing the chemical logic behind the method—not as a happy accident, but as a predictable outcome.
In closing, the new work by Assistant Professor Yuuki Mochizuki and Associate Professor Naoto Tsubouchi presents more than just a clever technique—it is a new controlled thermochemical strategy for lithium recovery that’s both elegant and grounded in chemical intuition. They successfully showed that dry processing can not only rival but in many cases outperform traditional hydrometallurgical methods in terms of both selectivity and simplicity. What makes this particularly relevant is how well it fits the realities of large-scale battery recycling: limited chemical waste, fewer processing stages, and compatibility with high-temperature industrial setups already in use. In a context where lithium demand is surging and raw material supply chains are growing more fragile—geopolitically and environmentally—the ability to isolate lithium efficiently, without co-extracting nickel, cobalt, or manganese, becomes critically important. Their method does this without the usual chemical excesses—no acids, no solvents, no complex leachate management. That alone sets it apart. And yet, what’s most striking isn’t just what the process avoids, but how deliberately it works with the inherent chemistry of the materials rather than against it. Choosing to oxidize metal chlorides into stable oxides while allowing lithium chloride to volatilize isn’t just a practical decision—it reflects a deeper alignment with the principles of green chemistry. It reduces intervention. It avoids unnecessary reagents. And it results in solid residues that are far more straightforward to handle or reuse. There’s a clarity to the process that feels both efficient and, frankly, respectful of the materials themselves.
Importantly, the implications of this work extend beyond lithium alone. The approach hints at broader opportunities to design element-specific separations for other critical systems—especially where subtle thermodynamic differences can be exploited. Rather than treating spent materials as hopelessly tangled waste, this method encourages us to see them as recoverable layers, waiting to be cleanly peeled apart under the right conditions. We think the study is also impactful because it addresses sustainability without complicating the industrial landscape. For researchers, it suggests new terrain worth exploring. For manufacturers, it offers a viable alternative to increasingly strained supply chains. And for the broader shift toward circular materials use, it’s a reassuring reminder that well-designed chemistry still has a lot to offer.
Reference
Mochizuki, Yuuki & Tsubouchi, Naoto. (2025). Selective Separation of Li, Ni, Co and Mn from Model Spent Li Ion Battery Cathode Materials by Dry Processing using the Combination of Chlorination and Oxidation. Reaction Chemistry & Engineering. 10. 10.1039/D4RE00328D.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



