Significance
Synthetic polymers (plastics) are an essential part of modern-day economy and have found many uses ranging from house hold to industrial scale. However, the ever-increasing production and the poor degradability of plastics have raised significant environmental concerns both in the oceans and on land. By definition, plastic pollution is the accumulation of plastic objects and particles (micro-, meso- or macro debris) in the Earth’s environment that adversely affects wildlife, marine life, wildlife habitat and humans. Consequently, measures to avert a total environmental choke and reverse the current unpleasant environmental plastic conditions have seen researchers attempt the development of sustainable polymers that can be chemically recycled. As of now, the idea of chemical recycling presents the most readily available actions to address the aforementioned shortfalls. Ideally, the popular strategy for chemically recycling polymers is a polymer-to-monomer approach.
Noteworthy studies have established that many types of polymer, including poly(carbonate) (PC), polyester, and polythioester, can be quantitatively depolymerized to the corresponding monomers (or other small molecules), which can be potentially be reused to prepare the corresponding virgin plastics. However, for the polymer-to-monomer recycling process, sizable energy inputs are often required for additional operations such as separation and purification of monomers for subsequent polymerization; this is therefore a limitation.
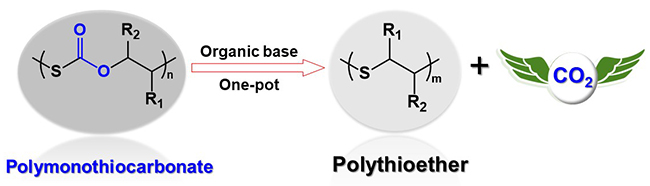
Alternatively, another promising approach is to recycle a polymer by transforming it to another one, i.e., the polymer-to-polymer method. This technique has the potential to transform polymers into closely related derivatives for the ultimate formation of high-value polymers; yet, absent from literature is relevant reports on this. To address this, researchers from the Department of Polymer Science and Engineering at Zhejiang University in China: Dr. Xiao-Han Cao, Dr. Cheng-Jian Zhang, Dr. Jia-Liang Yang, Dr. Lan-Fang Hu and led by Professor Xing-Hong Zhang developed a new polymer-to-polymer method for recycling a sulfur-containing polymer, poly(monothiocarbonate) (PMTC), to poly(thioether) in a one-pot/one-step process without the addition of other monomers. Their work is currently published in the research journal, Polymer Chemistry.
The researchers started with the degradation of PMTC to a cyclic monothiocarbonate that generated an episulfide via a decarboxylative reaction. Next, the episulfide was subjected to ring-opening polymerization. The researchers then used organic bases to catalyze the degradation of PMTC and initiate the ring-opening of the generated cyclic monothiocarbonate. This process was successfully expanded to transform several PMTCs to the corresponding poly(thioether)s.
The authors reported that the resultant poly(thioether)s exhibited higher thermal decomposition temperatures (up to 311 °C), higher refractive indexes (ca. 1.62) and lower glass transition temperatures (−48 °C) than those of the corresponding PMTCs. In addition, the team noted that the generated poly(thioether)s had significantly improved thermal properties in comparison with the corresponding PMTCs.
In summary, the study demonstrated a novel pathway for repurposing hydroxyl-terminated PMTCs to poly(thioether)s which was catalyzed by commercially available organic bases. The utility of a variety of organic bases and COS-derived PMTCs confirmed the versatility of the presented approach. Remarkably, the researchers demonstrated the potential of the generated poly(thioether)s as optical materials. In a statement to Advances in Engineering, Professor Xing-Hong Zhang further emphasized that their approach was vital for the upcycling of PMTCs and better still, met the requirements of sustainable chemistry.
Reference
Xiao-Han Cao, Cheng-Jian Zhang, Jia-Liang Yang, Lan-Fang Hu, Xing-Hong Zhang. Repurposing poly(monothiocarbonate)s to poly(thioether)s with organic bases. Polymer Chemistry, 2020, volume 11, page 309.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



