Significance
The status of the renewable energy situation necessitates efficient energy storage systems that can manage these volatile resources. Recently intensive research has been conducted on large-scale electrical energy storage because such technologies promise be an energy game-changer. As it stands, today’s minimal efforts for energy storage are not sufficient for leveraging excess renewable energy.
Electrolysis is a technique that uses a direct electric current to drive an otherwise non-spontaneous chemical reaction. It can be used to store electrical energy by splitting water into hydrogen and oxygen; i.e. products that contain chemically free energy. Hydrogen is quite satisfactory on small scale applications but methane is more preferable, reason being that the former has less enthalpy per volume when compared to the latter. Also, methane can be directly introduced into existing pipe systems. Electrolysis to achieve methane can be done in two ways: first, via commercial water electrolysis and second, via CO2-water co-electrolysis. In view of recent research trends towards intermediate temperature direct electrolysis, Caesium dihydrogen phosphate (CsH2PO4) when used as an electrolyte with nickel cathode, presents an alternative means of generating methane.
Previous reports showed several attempts to use CsH2PO4 as the electrolyte in intermediate temperature fuel cells, however, only few attempts have been made on water electrolysis and none under pressurized conditions and in the presence of CO2. Therefore, to address this, researchers from the Technical University of Denmark: Dr. Aleksey Nikiforov, Dr. I.M. Petrushina, Dr. Eric Christensen, Dr. Rolf W. Berg and Professor Niels J. Bjerrum reported the electrochemical behavior of CO2 on a Nickel cathode at higher temperatures and the dependence on the partial pressure of carbon dioxide during such an electrolysis. Their work is currently published in the research journal, Renewable Energy.
To begin with, the research team fabricated the quartz cells that they needed to study their system i.e. to investigate the electrochemical behavior of nickel-coated electrodes. Next, the team coated and verified the coating of the electrodes they were to use. The cells to be used were then filled and sealed. Overall, cyclic voltammetry was performed with a nickel electrode in molten CsH2PO4 at 350 °C and at approximately 25 bar of water pressure. The effect of adding carbon dioxide was also examined.
The authors mainly observed that even small amounts of CO2 (partial pressure below 0.2 bar at 350 °C, corresponding to 0.5 bar at 22 °C of a 5:1 mixture of N2: CO2) had a pronounced effect on the cyclic voltammetry curves. In fact, under the aforementioned conditions, the team reported that hydrogen formation was substituted by another process, i.e. methane formation.
In summary, the Danish study presented in an elegant way electrolysis experiments with nickel cathodes in CsH2PO4 at 350 °C with different gas additions and under an intrinsic water pressure of approximately 25 bar. Generally, the scholars reported that the CO2 content of the gas above the melt and the conditions had a very important influence on the nature of the formed electrolysis products. In a statement to Advances in Engineering, the authors highlighted their proposed 1-Step reaction may be used to convert CO2, H2O and electricity to methane and oxygen.
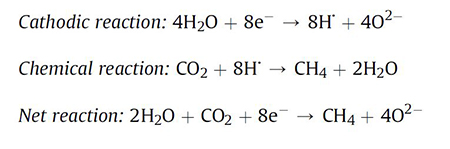
Reference
A. Nikiforov, I. Petrushina, E. Christensen, R. W. Berg, N. J. Bjerrum. Voltammetric study of one-step electrochemical methane production during water and CO2 co-electrolysis in molten CsH2PO4. Renewable Energy, volume 145 (2020) page 508-513.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.


