Significance Statement
Nanoscaled zinc oxide used as a nanoscale photo-initiator has the capacity to initiate radical polymerization of bulk materials. However, can be sensitive to the preparation conditions. The developed photo semiconductor zinc oxide has the additional potential to be applied as photo-initiators for the development of multifunctional acrylic esters in bulk. For these commercially applied polymerization procedures for example for dental applications or food packaging, the migration as well as transport of molecular polymerization initiators appears to be a problem. Even the newest commercial polymeric initiators, as recently proven analytically, lead to potential health risks.
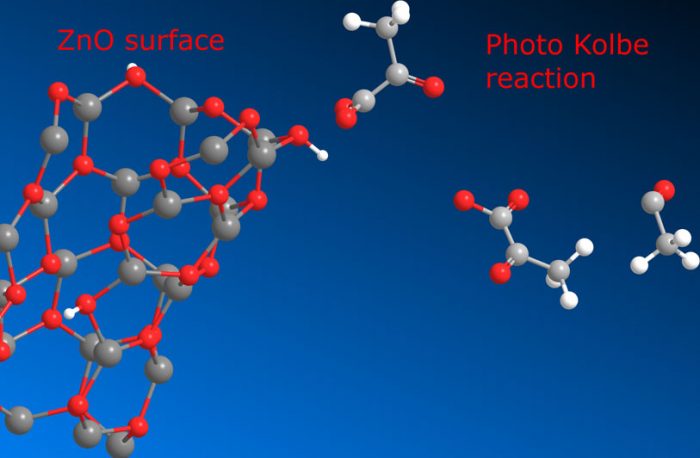
Michael Schmitt and Jacques Lalevée at Saarland University in Germany and at University of Haute Alsace in France investigated the Photo-Kolbe effect and zinc oxide synthesis and dealt with the other effect of the modifier content during the preparation. The originality of their study was founded on the presented modifier levulinic acid, which is a small molecule without reactive or aromatic functionalities. The synthesis process led to non-surface modified bare nanoparticles that were modified in the second step. Their work is published in Colloids and Surface A.
The authors therefore demonstrated the initiation potential of zinc oxide modified with non-photo-reactive levulinic acid for the polymerization of a multifunctional acrylic ester mixture. For this reason, the reaction was monitored in situ by a transmissions curing equipment sensitive to solidification of the system. The authors observed that time, the acidity of the reaction solution, and the total amount of modifier affected the results of the photo-polymerization. The authors also found that additional syntheses optimization were important to control the reaction solution and to avoid untamed aggregation owing to time implications.
Doping and combination with metal ions that was identified to improve the efficiencies of these systems by reducing the number of valence band electrons therefore affecting the lifetime of the holes, influenced the dispersion in the resin in a non-investigated way. The proposed procedure is therefore important for additional applications. Small non-surface modified nanocrystals with high mono-dispersity as well as high size uniformity could be implemented in optoelectronic application. The proposed green-fluorescing UV-absorbing materials can result in further applications that can justify the steep cost of such initiating systems if aspects of health are insufficient.
Reference
Michael Schmitt and Jacques Lalevée. ZnO nanoparticles as polymerization photo-initiator: Levulinic acid/NaOH content variation. Colloids and Surfaces A: Physicochemical and Engineering Aspects, Volume 532, 5 November 2017, Pages 189-194
Go To Colloids and Surfaces A
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.



