Nano Research, September 2014.
Juan Zhang, Ying Liu, Jun Lv, Genxi Li.
Laboratory of Biosensing Technology, School of Life Sciences, Shanghai University, Shanghai, 200444, China and
State Key Laboratory of Pharmaceutical Biotechnology, Department of Biochemistry, Nanjing University, Nanjing, 210093, China.
Abstract
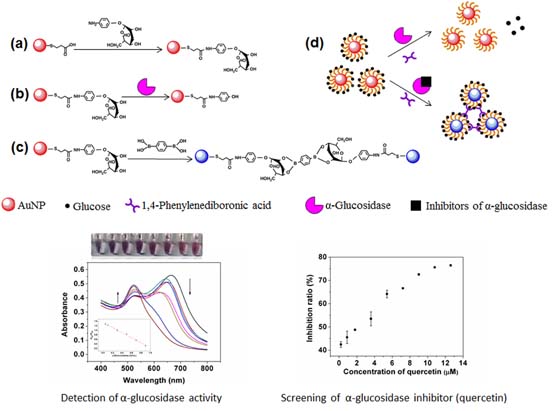
A colorimetric method has been established for {Alpha}-glucosidase activity assay and its inhibitor screening. The method is based on the specific recognition between 1,4-phenylenediboronic acid (PDBA) and 4-aminophenyl-{Alpha}-d-glucopyranoside (pAPG), which may induce aggregation of pAPG-functionalized gold nanoparticles (AuNPs) to achieve color change of the test solution. Because pAPG is the substrate of {Alpha}-glucosidase, the aggregation of AuNPs will be influenced by {Alpha}-glucosidase since there is no coordination reactivity between PDBA and 4-aminobenzene, the hydrolyzed product of pAPG catalyzed by the enzyme. Therefore, a simple and easily-operated colorimetric method for the assay of {Alpha}-glucosidase activity can be developed. Under the optimized experimental conditions, the ratios of absorbance at a wavelength of 650 nm to that at 520 nm vary linearly with the {Alpha}-glucosidase activity within a range from 0.05 to 1.1 U/mL with a lowest detection limit of 0.004 U/mL. Moreover, using the proposed method, the inhibition effect of gallic acid and quercetin on {Alpha}-glucosidase activity can be tested with IC50 values of 1.16 mM and 1.82 uM, respectively. Thus, the method has a great potential not only for the detection of {Alpha}-glucosidase activity, but also for the screening of its inhibitors.
Additional Information
A simple and yet effective colorimetric method has been firstly demonstrated for {Alpha}-glucosidase activity assay and the inhibitor screening. The method is based on the specific recognition between 1,4-phenylenediboronic acid (PDBA) and 4-aminophenyl-{Alpha}-Dglucopyranoside (pAPG), which may induce aggregation of pAPG functionalized gold nanoparticles (AuNPs) to achieve color change of the test solution. Because pAPG is the substrate of {Alpha}-glucosidase, the aggregation of AuNPs will be influenced by {Alpha}-glucosidase since there is no coordination reactivity between PDBA and 4-aminobenzene, the hydrolyzed product of pAPG catalyzed by the enzyme. Therefore, a simple and easily-operated colorimetric method for the assay of {Alpha}-glucosidase activity can be developed. Under the optimized experimental conditions, the ratios of absorbance at the wavelength of 650 nm to that at 520 nm are linear with the {Alpha}-glucosidase activity within a range from 0.05 to 1.1 U/mL with a lowest detection limit of 0.004 U/mL. Moreover, using the proposed method, the inhibition effect of gallic acid and quercetin on {Alpha}-glucosidase activity can be tested with IC50 values of 1.16 mM and 1.82 uM, respectively. The method bears advantages in the low technical and instrumental demands, wide detection range, and high sensitivity, and thus it will have a great potential for the detection of {Alpha}-glucosidase activity and the screening of the inhibitor.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.