Chem Asian J. 2013;8(9):2125-35.
Itoh S, Tokunaga T, Sonoike S, Kitamura M, Yamano A, Aoki S.
Production Technology Laboratories, Kaken Pharmaceutical Co. Ltd. 301 Gensuke, Fujieda, Shizuoka 426-8646, Japan.
Abstract:
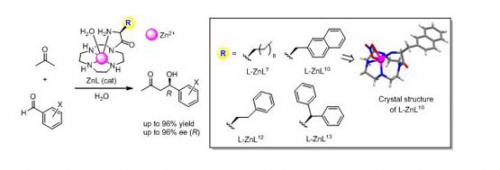
The aldol reaction is one of the most important C–C bond forming reactions, and a wide variety of efficient asymmetric catalysts have been developed. In natural methabolic pathway in living systems, aldol reactions are catalyzed by aldolases enzymes in stereospecific and reversible manners. In class I aldolases, an enamine intermediate is formed between a lysine residue of the enzyme and the carbonyl group of the substrate. In class II aldolases, a zinc(II) ion cofactor acts as a Lewis acid, with enolates being generated at the active site. The majority of the reported organocatalysts described above function as class I aldolase mimics. On the other hand, several mimics for class II aldolases have been reported.We previously reported that chiral Zn2+ complexes, which are designed to mimic the action of class I and II aldolases, catalyzed enantioselective aldol reactions of acetone and analogues thereof with benzaldehyde derivatives. In this study, we report on the synthesis of new chiral Zn2+ complexes having Zn2+ tetraazacyclododecane (Zn2+–[12]aneN4) and amino acid parts containing aliphatic, aromatic, anionic, cationic, and dipeptide side chains. The chemical and optical yields of the aldol reaction were improved (up to 96% ee) by using ZnL complexes of L-decanylglycyl-pendant [12]aneN4 (L-ZnL7),L-naphthylalanyl-pendant [12]aneN4 (L-ZnL10),L-phenylethylglycyl-pendant [12]aneN4 (L-ZnL12), andL-biphenylalanyl-pendant [12]aneN4 (L-ZnL13). UV/VIS and CD (circular dichroism) titrations of acetylacetone (acac) with ZnL complexes confirmed that a ZnL–(acac)– complex is exclusively formed and not the enaminone of ZnL and acac, as we previously proposed. An X-ray crystal structure analysis of L-ZnL10 and L-ZnL13 show that the NH2 groups of the amino acid side chains of these ligands are coordinated to Zn2+ as the fourth coordination site, in addition to three nitrogens of [12]aneN4 rings.These data indicate that the Zn2+-bound OH– of ZnL complexes (ZnL(OH–)) acts as a base to deprotonate the {Alpha}-proton of acetone to generate the ZnL–(enolate)– complex in these aldol reactions, rather than our previous hypothesisdescribed in our previous paper (Chem. Eur. J. 2009), in which the amine group in the side chainwas hypothesized to act as the base.These results should afford useful information concerning the reaction mechanism of aldol reaction catalyzed by artificial Zn2+ catalysts and should extend our knowledge of the mechanism of action of class II aldolases.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.