Angewandte Chemie International Edition, Volume 52, Issue 6, pages 1731–1734, February 4, 2013.
Dr. Shenci Lu, Si Bei Poh, Woon-Yew Siau, Prof. Yu Zhao
Department of Chemistry, National University of Singapore, 3 Science Drive 3, Singapore 117543 (Singapore) http://zhaoyu.science.nus.edu.sg
Abstract
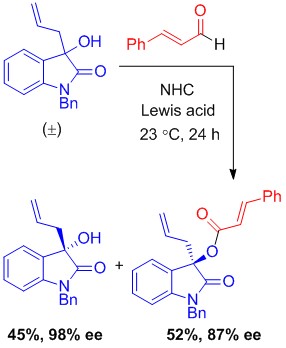
Enantioselective: The first highly enantioselective kinetic resolution of 3-hydroxy-3-substituted oxindoles has been developed through oxidative esterification catalyzed by a N-heterocyclic carbene (see picture). This method uses a simple procedure and provides 3-hydroxy-oxindoles with various substituents at the 3-position in excellent enantiopurity. S=selectivity.
Copyright © 2013 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
Additional Information
The ubiquity of alcohol functionality in natural products and pharmaceuticals makes their enantioselective preparation an important goal in synthetic organic chemistry. In contrast to the well-established kinetic resolution of secondary alcohols, the synthetic utility of tertiary alcohol kinetic resolution has remained very limited. Recently we have reported in Angewandte Chemie (2013, 52, 1731-1734) the first highly enantioselective catalytic kinetic resolution of oxindole-derived tertiary alcohols, which are important targets and intermediates in medicinal chemistry. The high level of differentiation of the two tertiary alcohol enantiomers (selectivity factor up to 78) has been realized through oxidative esterification catalyzed by chiral N-heterocyclic carbene (NHC), with the aid of cooperative catalysis by Lewis acid.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.