Chemical Engineering Journal, Volume 260, 15 January 2015, Pages 399–410.
Diego H. Quiñones1, Pedro M. Álvarez1, Ana Rey1, Sandra Contreras2, Fernando J. Beltrán1, .
- Departamento de Ingeniería Química y Química Física, Facultad de Ciencias, Universidad de Extremadura, Av. de Elvas s/n, 06071 Badajoz, Spain and
- Departament d’Enginyeria Quimica, Universitat Rovira i Virgili, Campus Sescelades, Av. Països Catalans 26, 43007 Tarragona, Spain.
Abstract
Aqueous mixtures of six commonly detected emerging contaminants (acetaminophen, antipyrine, bisphenol A, caffeine, metoprolol and testosterone), selected as model compounds, were treated by different solar-driven photochemical processes including photolysis, photocatalytic oxidation with Fe(III) or TiO2, photo-Fenton and single, photolytic and photocatalytic ozonations. Experiments were carried out in a compound parabolic collector photoreactor. It was found that photolysis and photocatalytic oxidation using Fe(III) are not effective for the complete removal of the selected contaminants, while TiO2 photocatalysis, photo-Fenton, single, photolytic and photocatalytic ozonations can rapidly remove them and decrease total organic carbon to some extent. The combination of photocatalytic oxidation and ozonation considerably enhances the system efficiency by reducing the ozone demand and energy requirements to completely remove the contaminants. Results also demonstrate that, at the operational conditions applied in this work, the contaminant removal and mineralization by ozone processes takes place in the slow kinetic regime, therefore the application of the ozone combined processes studied instead of single ozonation is recommended. Kinetic considerations on the application of solar photocatalytic processes for mineralization have been also assessed.
Go To Chemical Engineering Journal
Significance Statement
Solar photocatalytic ozonation, which makes simultaneous use of ozone, solar radiation and catalysts, is introduced as novel and more efficient water treatment process. This technology gives rise a synergistic effect that leads to faster and complete pollutant removal from contaminated water and, therefore, enhances the efficiency of single tertiary treatments such as photocatalytic oxidation and ozonation.
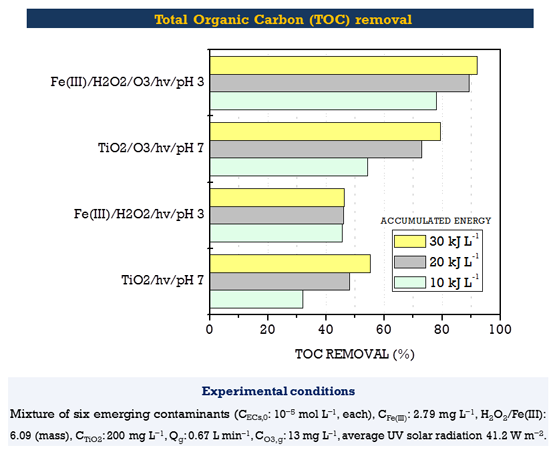
When applying photocatalytic ozonation processes assisted by photo-Fenton and titania in a solar pilot scale experimental system, a complete removal and high mineralization of a mixture of several emerging organic compounds was achieved. The selected model compounds, which are attributed to negative effects such as genetic mutation, carcinogenesis, birth defects and endocrine disruption, were recalcitrant to degradation by photolysis and iron assisted photocatalytic oxidation. Single ozonation, TiO2 based photocatalysis and photo Fenton processes lead to the complete removal of the contaminants but no important mineralization is achieved after 5 hours of treatment.
Additionally, photocatalytic ozonation processes are interesting alternatives since they were found to make a more efficient use of ozone to mineralize total organic carbon by diminishing the ozone requirements and treatment times.
The significance of the present work lies in exploring effective alternatives to degrade priority and emerging contaminants in water that are recalcitrant to conventionally applied biological and physicochemical wastewater treatment processes. Sunlight was used as UV radiation source to overcome the high operative costs that the use of UV lamps at industrial scale would generate. This work is also important since so far very few relating the application of solar photocatalytic ozonation at pilot scale have been published.
 Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.
Advances in Engineering Advances in Engineering features breaking research judged by Advances in Engineering advisory team to be of key importance in the Engineering field. Papers are selected from over 10,000 published each week from most peer reviewed journals.